C 12 atom11/22/2023 If they were to exist there permanently, the Heisenberg uncertainty principle would predict their momentum to be at least 1^-20 kg m/s^2, which corresponds to a kinetic energy of about 20 MeV, considerably higher than the energies of the electrons emitted in beta-decay. However, we now know that electrons are created inside the nucleus only at the moment of beta-decay. Rutherford thought the neutron was a combination of two particles: a proton and an electron, since electrons are emitted from nuclei in beta-decay reactions. This radius can also be represented as 20 fm, where 1fm = 10^-15m, the femtometer or fermi unit. We have seen how Rutherford estimated the radius of a silver nucleus to be 2 x 10^-14 m, by observing the angular dependence of alpha-particle scattering. The fact that C-12 nucleus has a mass slightly less than that of its 12 constituent particles indicates that some mass is lost (as energy, E=mc^2) when the particles are combined into a nucleus. The mass of an isolated proton has been measured to be 1.0073u and that of a neutron is 1.0087u (about 0.14% larger). Natural carbon consists mainly of the A=12 isotope (carbon-12) but contains 1.1% of carbon-13, so the atomic weight of carbon is actually 12.011, which represents a weighted average over all the isotopes present.īy definition, the mass of the carbon-12 nucleus is 12u, where u is the atomic mass unit (amu), equal to 1.66 x 10^-27 kg. Hydrogen normally has Z=1, A=1 but can be made with A=2 (deuterium) and A=3 (tritium). Although Z is always the same for a given element, the number of neutrons (and therefore A) is found to vary, giving rise to different isotopes of the element. Together, these two kinds of particles, when present within a nucleus, are called nucleons and their total number A is the mass number of the nucleus, which is closely related to the atomic weight of the corresponding element. 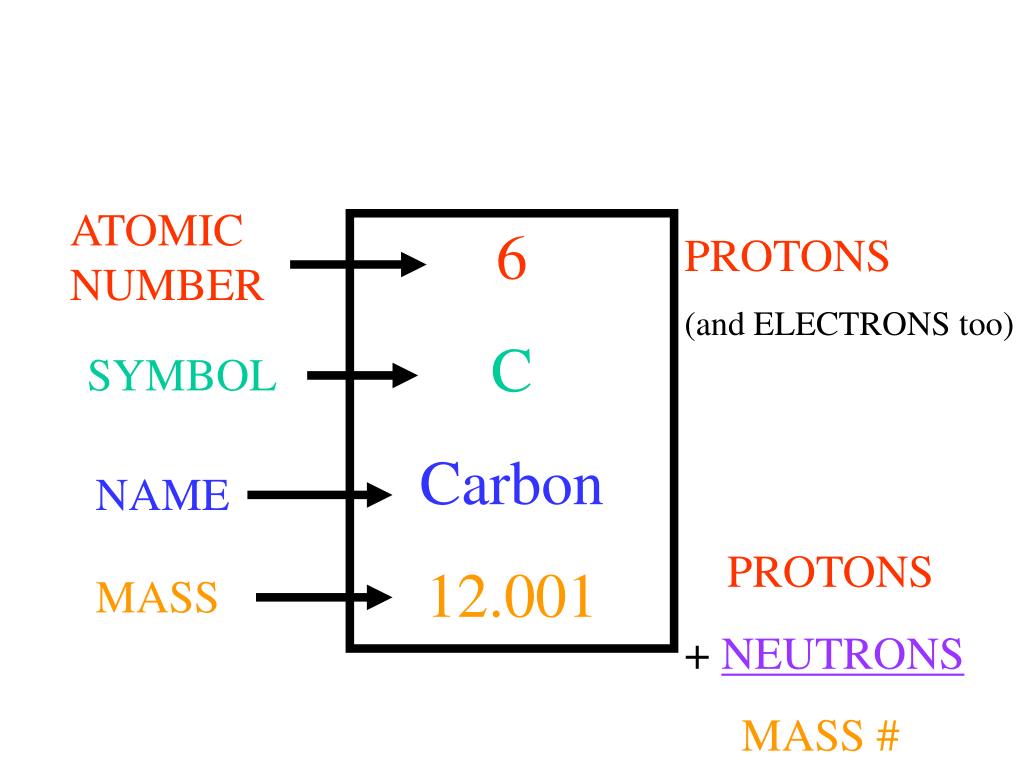
We now know that an atomic nucleus contains Z protons (Z being the proton number of the atom, equal to the atomic number of the corresponding element), each of which has a positive charge (+e), and N uncharged neutrons (N being the neutron number). Two years later, Marie Curie named this emission of radiation from atomic nuclei radioactivity, and less 20 years later Rutherford had used this radiation to investigate properties of the atom and its nucleus (including the nuclear size). Our knowledge of nuclear physics started with Becquerel's accidental discovery (1896) that crystals of a uranium salt emit radiation which blackens a photographic emulsion. Since the nucleus is very small (the particles it contains are highly confined), the Heisenberg uncertainty principle tells us that the momentum uncertainly will be large, the energy levels far apart and the energies involved in nuclear reactions correspondingly large (typically MeV).  
Wave mechanics can be applied to the nucleus of an atom, as well as to its surrounding electrons. Figure references are to the second edition of Modern Physics by Serway, Moses and Moyer (Saunders, 1997)
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
